Traditional mammalian cell culture is usually anchor-dependent and serum-supplemented, which is directly connected to a series of disadvantages, due to the serum/ animal derived components. In contrast, suspension cultures in animal-derived component free (ADCF) cell culture media have several advantages:
- reduced lot-to-lot variability of the medium
- easier scale-up
- no risk of blood-borne pathogens
- massively reduced production costs
- increased animal-welfare
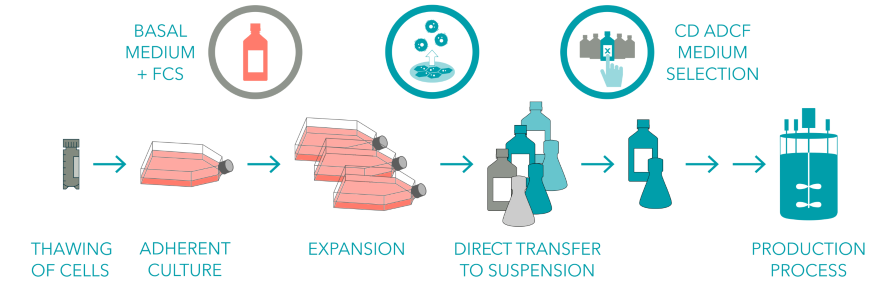
Infographic depicting general steps in the cell line adaptation process.
Xell’s scientists have more than 20 years of experience with cell culture and state-of-the-art culture media. Adapting cell lines to serum-free suspension culture is just one of the many services we offer to the scientific community and biotechnology industry. Contact us to discuss your specific project.






